
The radiocarbon dating method, the most relied-upon scientific dating tool used in archeology today, estimates the ages of organic materials like wood charcoal, bones, basketry, by measuring the quantity of unstable cosmic isotope radiocarbon. The dawn of radiocarbon dating in the late 1940s and 1950s upset archeological chronologies around the world; time and time again, things and cultures proved to be older (or younger) than experts had reasoned. Today’s archeologists routinely send off samples to be dated, yet radiocarbon dating remains poorly understood by many archeologists. This exhibit explains the intricacies of radiocarbon dating to science lovers and serious students of archeology to help guide the wielding of this powerful tool.
Radiocarbon dating is complex—it interweaves physics, chemistry, and biology. Its archeological application often requires an understanding of geology as well as archeology. Further, radiocarbon scientists are constantly making methodological and theoretical innovations. The savvy archeologist must grasp not only how radiocarbon dating works, but also keep up to date with best practices and understand how radiocarbon dating was undertaken and interpreted by earlier archeologists.
This exhibit is a technical overview that requires a basic familiarity with chemistry and physics. Herein lies explanation of the fundamentals of radiocarbon dating, from how to understand calibration curves, to developing a dating strategy, selecting samples, reporting radiocarbon data, evaluating the utility of a given radiocarbon assay, building more sophisticated chronologies, and leaving a legacy of high-quality radiocarbon data for future researchers.
Scientific Literacy Test?
This special exhibit on Radiocarbon Dating may test your scientific literacy. The most critical technical terms and essential radiocarbon concepts are explained herein. In-depth writing about a complicated technique requires the use of many specialized terms that scientifically literate readers would likely know, and some they may not. Fortunately, with a simple web search you can bone up quickly on peculiar terms like “Faraday cup” on Wikipedia or other authoritative sources.
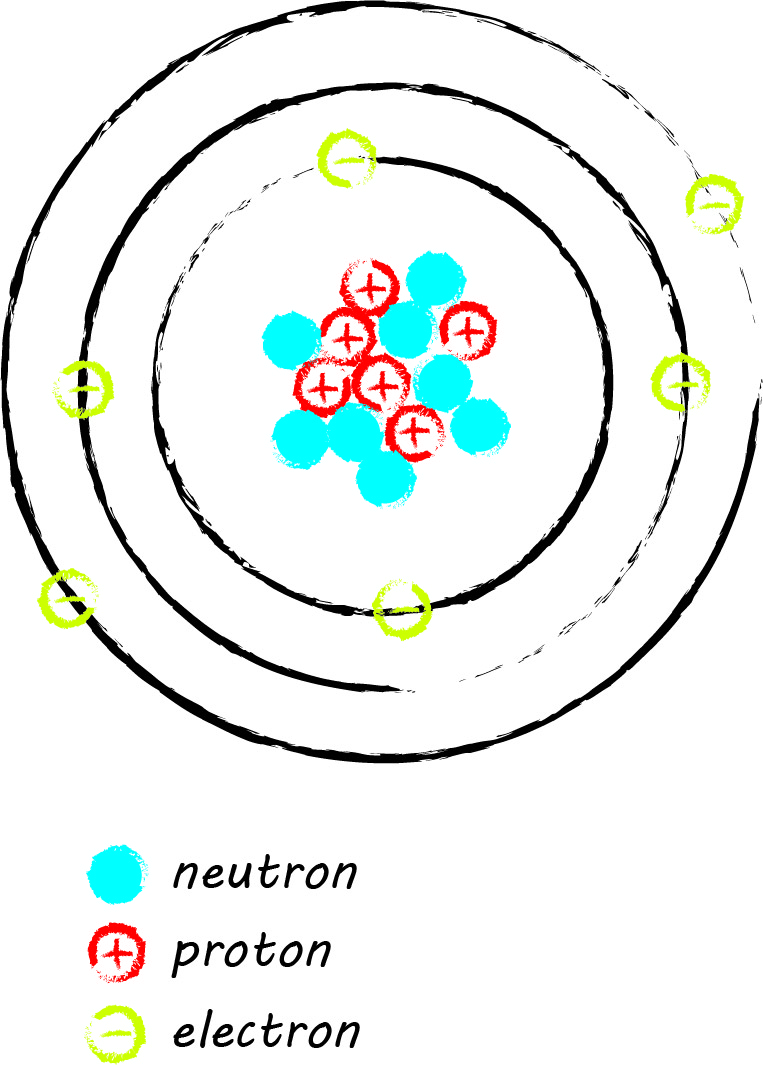
Radiocarbon (scientific shorthand: 14C) is an unstable isotope of carbon. Like the stable carbon isotopes, it is in every living organism. Look at your hand; your skin, bones, fingernails, and hairs all contain carbon. Cosmic processes in earth’s upper atmosphere create radiocarbon, which reacts with oxygen and becomes carbon dioxide as it is distributed around the planet (more on this in Dating 101). During photosynthesis carbon dioxide is incorporated into plants, and the carbon is conveyed to animals who eat the plants, and so on up the food chain. Upon death, radiocarbon decays without replenishment.
As the radiocarbon in the dead organic material (plant or animal tissues) decay over the millennia, the quantity eventually becomes so little that our modern measurement technologies cannot detect it. For this reason, only materials which were alive during the last 55,000 years or so can be radiocarbon dated. Importantly, radiocarbon dating is a probabilistic tool which estimates a date range for when organisms died or became separated from the environment—it does not provide an exact date! Multiple assays and statistical methods can sometimes substantially narrow the likely date range.

A sample of material that has been radiocarbon dated is sometimes referred to as a radiocarbon assay; to assay is to measure or investigate the component of some material, in this case, carbon isotopes in archeological organic materials. Commonly assayed archeological materials include wood charcoal, bone, and shell, though almost any biological material can be dated. Some material types from certain archeological contexts require specialized treatment in the lab or corrections applied after radiocarbon measurement. All interpretations of radiocarbon assays require careful evaluation of how the context and dated event relate.
Since the onset of radiocarbon dating shook up culture chronologies developed in the first half of the 20th century, it has remained a critical archeological tool, spurring methodological and theoretical development across the discipline. Physicists, chemists, and archeologists specializing in radiocarbon dating continue the radiocarbon revolutions, as the invention and subsequent improvements of the method are often referred.
Though radiocarbon dating methods remain poorly understood by many, the "magic black box" mentality to radiocarbon dating is waning. In part, this is thanks to pioneering archeologists who have recognized that the sheer number of radiocarbon assays made over the decades have great potential to address large-scale research questions about the human past through statistical methods. The growing interest among archeologists in modelling chronological information using large radiocarbon data sets has refocused attention on the necessity of well-reported and critically interpreted radiocarbon data.

In This Exhibit
The following sections will guide students of archeology through the intricacies of radiocarbon dating. Begin with Dating 101 for an overview of key terms and radiocarbon concepts, including radioactive decay, 14C measurement, and calibration.
In the Environment discusses how 14C behaves in earth's atmosphere, terrestrial environments, and marine environments. In this section, learn about how 14C is created and distributed, why calibration curves are necessary, how environments store carbon differently (reservoir effects), how photosynthesis affects distributions of carbon isotopes (isotopic fractionation) in plants, and how 14C moves through the food chain. In the Lab reveals what happens in modern radiocarbon pretreatment, graphitization, and Accelerator Mass Spectrometer (AMS) measurement labs. Step back in time to the fascinating early history of the invention of radiocarbon dating with In Libby's Test Tube. The histories of other radiocarbon breakthroughs—the development of calibration curves in the late 1950s and 1960s, and radiocarbon dating by AMS in the late 1970s and 1980s—are also discussed here.
Selecting Samples reviews factors to consider when choosing radiocarbon samples. Reporting Results is a practical, and critical, read for people who use radiocarbon data. It explains how radiocarbon measurements are reported by the lab and discusses the most essential data for publication. Interpreting Dates discusses how to think critically about radiocarbon data and how they can be used to address research questions. Visit the Case Study: Radiocarbon Dating the Lower Pecos Canyonlands for a real-world example of how a large set of radiocarbon data can be used to address regional research questions. The concluding section, Radiocarbon Takeaways summarizes important points made in the exhibit and highlights research opportunities in this twenty-first-century age of "big data." Play learning games for kids of all ages on TBH Kids: Radiocarbon Dating Games. For further reading and source information see Credits & Sources.